Table of Contents
Sophie James, co-founder and CEO of Anomer Bio, sat down with Onyx for a feature-length interview on modified monosaccharides, intracellular glycan processing, and a rare-disease-first route into fibrosis, oncology, and Alzheimer's.
Could you tell us a bit more about Anomer Bio - what exactly are you working on, and how is it benefiting patients?
My now co-founder, Marcos, and I started our PhDs at the same time, both partly at Barts Cancer Institute. We bumped into each other when I was exploring entrepreneurship, figuring out where I wanted to go. We went to the pub after an event, and the next day Anomer Bio started. It was a whirlwind from there. You know what they say - the best ideas start in the pub.
We started building this alongside our PhDs. We'd both been working in oncology: I was understanding why some drugs fail, and he was working more on the drug development side, with immunotherapies and preclinical testing for a variety of different therapeutic modalities. What we realised was twofold. The first key part is that a lot of drugs target proteins, but a crucial layer that is overlooked are the glycans that cover most proteins. These glycans - complex sugar structures - are what drive cellular communication between cells, but also how tissues are formed. Marcos was working with lots of biotech startups, he developed a preclinical testing platform, and we wanted to approach this in a different way.
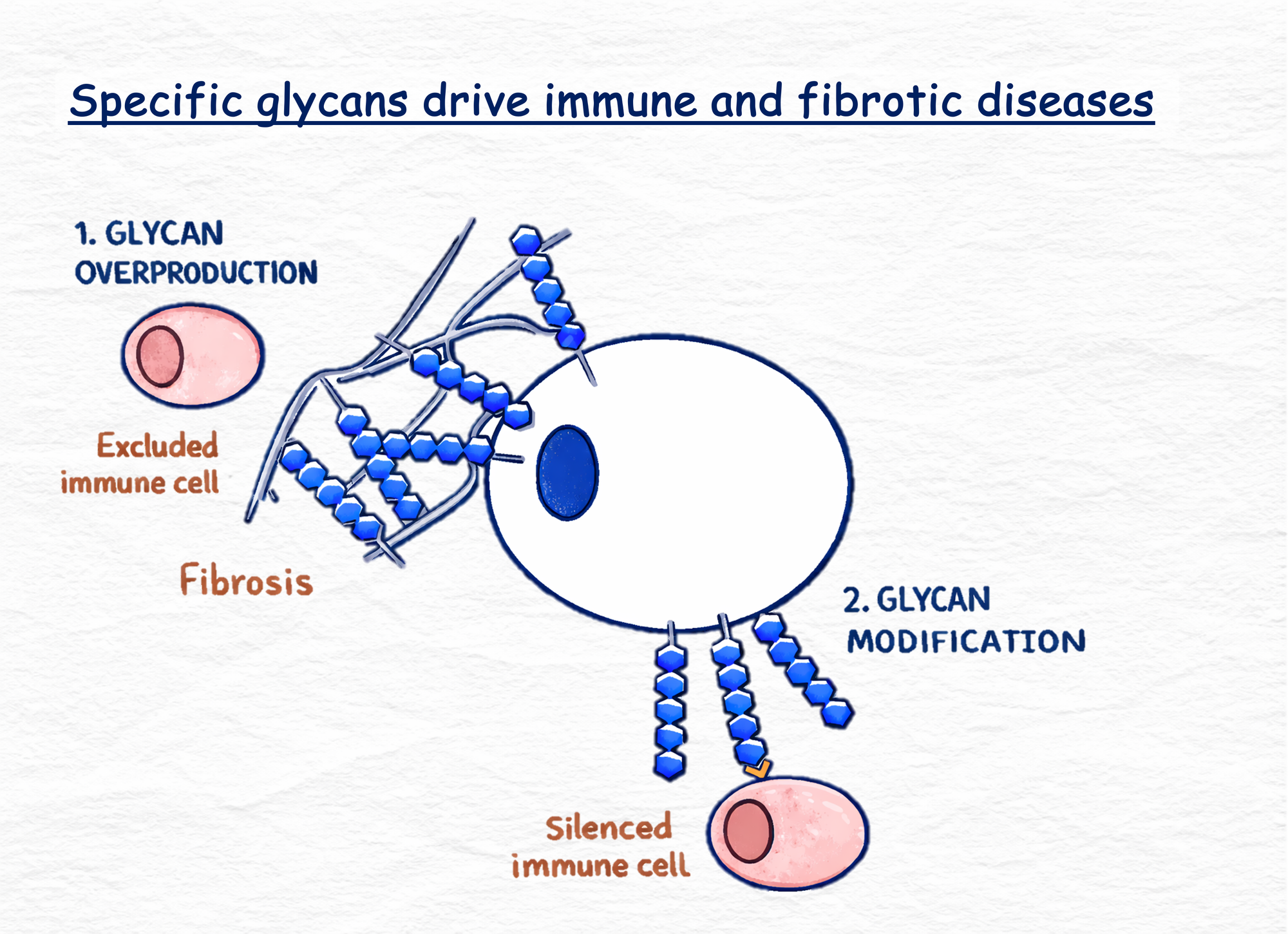
The inspiration for what we're doing - we're making small molecule drugs that change how proteins are glycosylated across immunofibrotic diseases - came from early work in the 1990s by Dr. Werner Reutter, which showed that the precursors for these complex sugar structures, called monosaccharides, could be subtly modified: the enzymes that process these precursors could tolerate subtle differences in chemical structures, meaning you could modify these building blocks and produce a slightly modified full glycan at the surface. This was the basis for Carolyn Bertozzi's now Nobel Prize-winning research on chemistry in living cells. She was using modified monosaccharides as click probes that are taken up by cells to form a modified structure that chemical reporters could be clicked onto. She used this for imaging of glycans, which led to a whole range of discoveries, a lot of them around hypersialylation of cancer tumours, which is now being translated into various companies through different modalities.
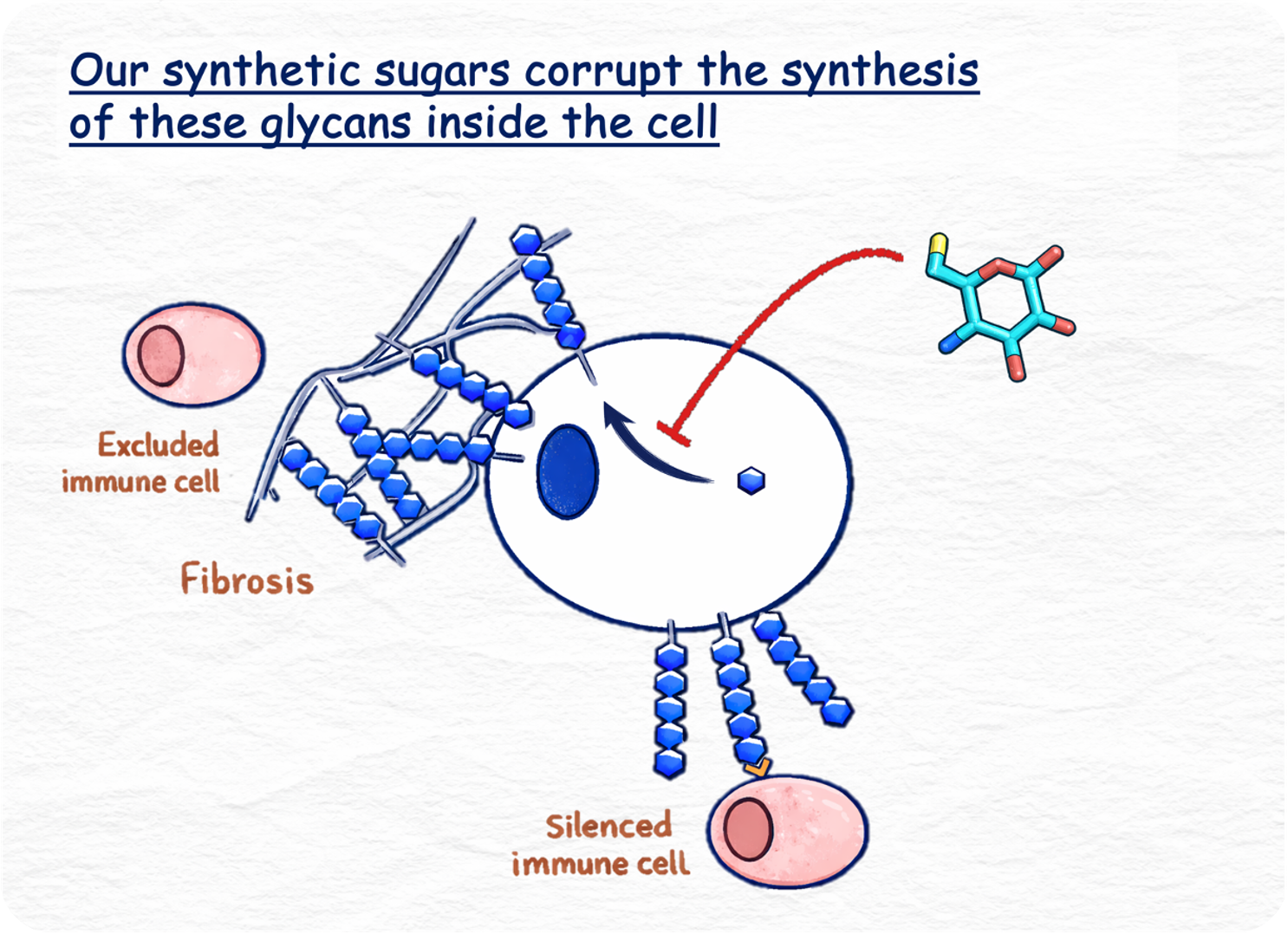
The reason I'm saying all of this is because the application of that chemistry to the biological problem is something we wanted to build on. The modified monosaccharide approach is the approach we are taking, but we're making it into a therapeutic rather than a research tool. Our drugs are small molecule, chemically modified monosaccharides that form a modified glycan at the cell surface and then overcome some of these disease-driving interactions. This is really around how tissues are structured and how they interact with the immune system.
And a bit about your own career background before starting the company?
My background is very scientific. I studied biology, and I've always been impact-driven and wanting to make a difference. That ranged a lot - everything from fieldwork in the rainforest in Indonesia to a placement in drug development at GSK. It was my time at GSK that reinforced my desire to work on people and healthcare. I really enjoyed working on the drug development process, and I learned a lot.
Based on that, I did my PhD in a clinical, translational lab in London. I thought this was where I could make a big difference. My PI was a consultant at Barts Hospital, and my whole PhD was trying to understand why some patients respond to targeted therapy in acute myeloid leukemia and why some patients don't. I was looking at a drug called gilteritinib, investigating metabolic mechanisms of resistance in the bone marrow. I enjoyed it and learned a lot, but I realised that academia is rewarded by publications, and progress can sometimes be slow. I wanted to see a translation from my research into the clinic.
At the same time, I became very interested in biotech and entrepreneurship, and I realised that this could be the answer. I started exploring. My PhD was at the Francis Crick Institute and Imperial, both very translational places. I started going to events, and I won a pitching competition in 2024 and began speaking to investors. That was the trigger for thinking this is what I want to do - it matches my skill set and my motivations. And it all started from there.
So what types of indications are you going for first?
We see ourselves very much as a glycan-targeting company with a platform - beginning with in silico and through to validation - that designs and screens small molecule glycan-targeting drugs. The indication we're going after first is a rare childhood disease, where the glycan we're going after is specifically the pathological driver of the disease. It's caused by a dysfunctional enzyme that usually breaks down the glycans, so there's too much of the glycan all over the body.
This being a rare disease benefits from fast application to the clinic and accelerated regulatory approval. But crucially, it's the fastest place to get clinical modality validation in a disease where there's no target risk. Then we can use that validation - those same IND packages - to move into larger disease indications like fibrosis, oncology, and Alzheimer's.
We have some grants and collaborations at the moment, so while in-house we're focusing on asset development in the rare disease setting, we're building out target validation alongside it in other diseases, which is an exciting strategy. When we started the company, a lot of the value we built was on proprietary in silico models that we built to design and screen the glycan-targeting drugs. Since moving into the company full-time in September 2025, we have moved from in silico screening hits to in vitro validated drug candidates.
So am I right in thinking there are some mechanistically adjacent competitors? People working further downstream, looking at glyco-immune checkpoints and that kind of thing. You're going a little earlier upstream - blocking the synthesis of glycans rather than masking them or cleaving them. Can you talk about the decision for that approach and how you're different from competitors?
Absolutely. The most established target in the glycan-targeting field is, as you said, this immune checkpoint axis - the siglec–sialic acid axis. That is being targeted; just this week, Palleon Pharmaceuticals presented some of their data at AACR showing that their enzyme-antibody conjugates are showing a lot of promise, and they're moving into clinical trials in collaboration with a Chinese pharmaceutical company. So there is excitement in the field around these targets, and a lot of the approaches have been antibody-based and enzyme-based.
The key way that we are different is that because we're not relying on modifying extracellular glycans and extracellular interactions, we're able to target intracellular glycan processing pathways inside cells. We're not just limited to receptor–ligand interactions like the sialic acid axis. While that is still something we are going after, our approach through intracellular targeting opens up a whole new field of glycan targets that haven't really been translated into the clinic before.
This is what we see so much in the field - there is such solid data, literature, and foundations for how these glycans are implicated in so many diseases, but the translation clinically has just been so lacking. We think this is because there haven't been modalities that are able to go after this. Some of these glycans are about water retention, about tissue biophysics, and that's just not able to be targeted with some of the other modalities. We're really opening up an avenue with that.
Another thing to say is that even on the sialic acid–siglec avenue, these larger biological therapies - especially in solid tumours that are very dense - struggle to penetrate the tumour. With our small molecule approach - they're single sugar units - we're hoping that we'll see better tissue penetration in some of the more fibrotic indications as well.
From my understanding, the literature that supports your approach is actually some of the oldest compared to the newer approaches. So with this biosynthesis blocker approach, why do you think no one else has built a commercial programme over the past decade around this?
It's a question we get a lot, and I think it's multifaceted. There haven't been the tools to design and screen these structures. A typical chemistry approach for this problem would be to sit down, think about the different chemical groups that could be added, then synthesise them and test them - and that would take decades. With the way we've been able to leverage computational tools, we can do that much more quickly. The screening we've done in silico means we can refine a very big pool of compounds into a much smaller set that we can then synthesise and test. Some of these computational models are even helping on the synthesis side, working out the right synthesis pathways.
In terms of mapping the glycan pathways, and where modifying at certain stages would have an influence, that's also newer evidence and newer data that has come through - beyond just the glycan at the surface having this effect. It's continually increasing, and it's an interesting area to work in. While some of these mechanisms have been established for a long time, there's increasingly new data coming out about their implication in a further range of diseases. As clinical validation of targeting glycans continues, I think there will be much more of a push to try and translate this research into the clinic, where previously it was just recognised but known as difficult and very complicated.
My placement at GSK was trying to use an analytical technique to look at the glycosylation of antibodies. It's a big challenge, and we've had progress, especially on the glycosylation-of-antibody side - there are some cool technologies and companies being able to target that in a more precise way. But that's just one step, and actually targeting the glycans as the disease-driving target itself is something that's newly emerging. We think it's going to be the next wave of therapeutics in drug development.
On the machine learning side, when it comes to modelling cellular sugar processing, where is the training data coming from?
In terms of building our own in-house machine learning models, we're not making brand-new models. A lot of it is about how we use models that already exist - everything from AlphaFold to project protein structures, and then doing various screening and docking experiments on top. All of the data we use is from publicly available datasets that we interrogate in a unique way. That was deliberate, given we wanted to keep it separate from the university and didn't want to take IP from there, because we wanted not to be limited by the speed of that process.
So what does your roadmap look like over the next 12 to 24 months?
If I look back briefly, the last six months have been hectic. We only recently moved into the company full-time last September, and we spent three months in New York working with SOSV - formerly IndieBio. We achieved some exciting milestones and got some exciting traction in that time, including Innovate UK grants, a big collaborative academic grant, new advisors, and we won a pitching competition in Boston with 2048 Ventures.
The vision for the future is to maintain that momentum. We've raised about 50% of our pre-seed, and we're now looking to close that in the next month or so. The vision is to hire people - we're in those early stages now - and reach some more important milestones. That would be selecting a lead compound for IND-enabling studies in our lead indication, which is this rare disease; reaching important regulatory milestones in terms of orphan designation and interaction with the FDA; and building out the platform. As I said, we're already working on a CRUK grant to build out this validation in both oncology and fibrosis in terms of the targets, so then we can flip the asset development that we've done in the rare disease into these larger indications.
It's going to be a big 12 months. But I hope that this time next year we'll be raising our seed round with data that validates what we're doing in animals - with compounds that we can take forward as development candidates into the next stage of preclinical testing.
And what would you say is your main message to investors right now?
The key thing is that in such little time - our lab's only been open for about three months - we've already generated data that not only validates the computational models we built early on, but actually shows real promise for new drug candidates. So: back us now, back us early. We're moving very quickly, and we feel like this is going to open up a whole avenue in drug development that hasn't been explored before. It's so critical - we're pulling together all these different pieces: the core chemistry, the biological interactions, the literature that exists but hasn't been translated, and we're bringing that all together.








Comments